"Fossil evidence suggests that animals probably evolved sometime around 600 million years ago, and large animals appear around 575 million years ago. Animals have an absolute requirement for oxygen for their respiration, so it has often been speculated that large macroscopic animals (those with the largest oxygen requirement) evolved when oxygen rose to permissible levels, which would be about 10% of those levels we have today. Our study supports this scenario by showing that the first occurrence of large respiring organisms, some of which are likely stem-group animals, emerged on the Avalon Peninsula in Newfoundland in concordance with oxygenation of this local environment."
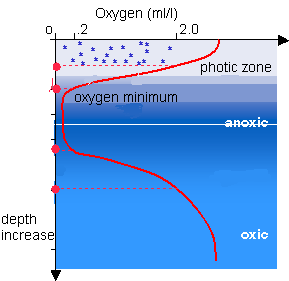 On Friday Don will be talking about Oxygen Minimum Zones. The Oxygen minimum zone (OMZ), is the zone in which oxygen saturation in seawater in the ocean is at its lowest. This zone occurs at depths of about 200 to 1,000 metres, depending on location. Surface ocean waters generally have O2 concentrations in equilibrium with the atmosphere. Colder waters can hold more oxygen than warmer waters. As this water sinks from the surface into deeper waters it is exposed to a rain of organic matter from above. As aerobic bacteria feed on this organic matter they use oxygen as part of the bacterial metabolic process lowering its concentration within the surrounding water. Therefore, the concentration of oxygen in deep water is dependent on the amount of oxygen it had when it was at the surface minus depletion by the degradation of organic matter. The OMZ relates to new ways scientists are looking at the 'Great Oxygenation Event'.
On Friday Don will be talking about Oxygen Minimum Zones. The Oxygen minimum zone (OMZ), is the zone in which oxygen saturation in seawater in the ocean is at its lowest. This zone occurs at depths of about 200 to 1,000 metres, depending on location. Surface ocean waters generally have O2 concentrations in equilibrium with the atmosphere. Colder waters can hold more oxygen than warmer waters. As this water sinks from the surface into deeper waters it is exposed to a rain of organic matter from above. As aerobic bacteria feed on this organic matter they use oxygen as part of the bacterial metabolic process lowering its concentration within the surrounding water. Therefore, the concentration of oxygen in deep water is dependent on the amount of oxygen it had when it was at the surface minus depletion by the degradation of organic matter. The OMZ relates to new ways scientists are looking at the 'Great Oxygenation Event'.The reading this week is another scientific paper which puts forward the idea the delivery of nutrients to the ocean following the last of the Snowball Earth Ice Ages (the Gaskiers glaciation) feed an algal (cyanobacterial) bloom that provided enough oxygen to fuel multicellular life - in other words animals. Don and his co-authors come up with this idea because the geochemistry of sediments before the Gaskiers glaciation is different from the geochemistry of the sediments afterwards.
Video primer about oxygen and photosynthesis
Wow. I would love to have a ocean named after me. I wonder if you can buy that like they do with stars? And how much would it cost? Could I just make a substantial donation to the U of M GeoSci Department and have the next great fossil they discover named the Moenckassorus? Also, having a wikipedia page is pretty cool too.
ReplyDeleteAs far as the substantive issues go, this seems to be related to a couple of the other lectures we've attended dealing with the formation of life on earth, how it evolved and what caused it to change. I would love to hear Dr. Canfield reference his dating techniques and his opinions on the extinction events that resulted in the end of the Canfield Ocean.
I was going to write the same thing as Nick; it would be sweet to have an ocean named after you. That being said, I've never heard of the Canfield Ocean until today.
ReplyDeleteI also think the evolutionary aspect is intriguing. I would like to see which oxygen-breathing species came about first and then look at the evolution of them all. It is interesting to think that all of the oxygen-breathing species arose only because the cyanobacteria started producing oxygen. I am curious about all of the species that came about due to this oxygen-rich environment.
What is a reasonable range for the reactive/total iron ratios? The paper states a minimum of 0.26 but doesn't say how high this value can get in anoxic oceans.
ReplyDeleteI told all my friends and family this weekend that I meet a guy who had a ocean named after him. Nobody thought it was very impressive but the important thing is that I think it is impressive. I am still a little confused about the sulfur relationship? What exactly did it demonstrate?
ReplyDeleteThis week's lecture was very interesting, especially for someone who lives close to the Marianas Trench.
ReplyDeleteMany learn of deep sea creatures such as the hanging-lantern-type fish, however, I wondered how they survive on the lowest levels of the ocean. If humans were to understand the natural processes of their low-oxygen lifestyle then perhaps ocean and space research could be more efficient.